Mature tertiary lymphoid structures predict immune checkpoint inhibitor efficacy in solid tumors independently of pd-l1 expression
- Select a language for the TTS:
- UK English Female
- UK English Male
- US English Female
- US English Male
- Australian Female
- Australian Male
- Language selected: (auto detect) - EN
Play all audios:
ABSTRACT Only a minority of patients derive long-term clinical benefit from anti-programmed cell death protein 1 (anti-PD-1) or anti-programmed death-ligand 1 (anti-PD-L1) monoclonal
antibodies. The presence of tertiary lymphoid structures (TLSs) has been associated with improved survival in several tumor types. Here, using a large-scale retrospective analysis of three
independent cohorts of patients with cancer who were treated with anti-PD-1 or anti-PD-L1 antibodies, we show that the presence of mature TLSs was associated with improved objective response
rates, progression-free survival and overall survival, independent of PD-L1 expression status and CD8+ T cell density. These results pave the way for using TLS detection to select patients
who are more likely to benefit from immune checkpoint blockade. Access through your institution Buy or subscribe This is a preview of subscription content, access via your institution ACCESS
OPTIONS Access through your institution Access Nature and 54 other Nature Portfolio journals Get Nature+, our best-value online-access subscription $32.99 / 30 days cancel any time Learn
more Subscribe to this journal Receive 12 digital issues and online access to articles $119.00 per year only $9.92 per issue Learn more Buy this article * Purchase on SpringerLink * Instant
access to full article PDF Buy now Prices may be subject to local taxes which are calculated during checkout ADDITIONAL ACCESS OPTIONS: * Log in * Learn about institutional subscriptions *
Read our FAQs * Contact customer support SIMILAR CONTENT BEING VIEWED BY OTHERS THE PD-1 EXPRESSION BALANCE BETWEEN EFFECTOR AND REGULATORY T CELLS PREDICTS THE CLINICAL EFFICACY OF PD-1
BLOCKADE THERAPIES Article 31 August 2020 PERIPHERAL CD4 MEMORY T CELLS PREDICT THE EFFICACY OF IMMUNE CHECKPOINT INHIBITOR THERAPY IN PATIENTS WITH NON-SMALL CELL LUNG CANCER Article Open
access 04 July 2023 TERTIARY LYMPHOID STRUCTURES AND B CELLS DETERMINE CLINICALLY RELEVANT T CELL PHENOTYPES IN OVARIAN CANCER Article Open access 21 March 2024 DATA AVAILABILITY The
datasets that support the findings of this study are not publicly available due to information that could compromise research participant consent. According to French/European regulations,
any re-use of the data must be approved by the ethics committee. Each request for access to the dataset (including the images) will be granted after a request sent to the corresponding
author (A.I.) and approval by the ethics committee. Source data are provided with this paper. REFERENCES * Vaddepally, R. K., Kharel, P., Pandey, R., Garje, R. & Chandra, A. B. Review of
indications of FDA-approved immune checkpoint inhibitors per NCCN guidelines with the level of evidence. _Cancers (Basel)._ 12, 738 (2020). Article CAS Google Scholar * Davis, A. A.
& Patel, V. G. The role of PD-L1 expression as a predictive biomarker: an analysis of all US Food and Drug Administration (FDA) approvals of immune checkpoint inhibitors. _J. Immunother.
Cancer_ 7, 278 (2019). Article Google Scholar * Cassetta, L. & Kitamura, T. Targeting tumor-associated macrophages as a potential strategy to enhance the response to immune checkpoint
inhibitors. _Front. Cell Dev. Biol._ 6, 38 (2018). Article Google Scholar * Schalper, K. A. et al. Elevated serum interleukin-8 is associated with enhanced intratumor neutrophils and
reduced clinical benefit of immune-checkpoint inhibitors. _Nat. Med._ 26, 688–692 (2020). Article CAS Google Scholar * Sautès-Fridman, C., Petitprez, F., Calderaro, J. & Fridman, W.
H. Tertiary lymphoid structures in the era of cancer immunotherapy. _Nat. Rev. Cancer_ 19, 307–325 (2019). Article Google Scholar * Ladányi, A. et al. Prognostic impact of B-cell density
in cutaneous melanoma. _Cancer Immunol. Immunother._ 60, 1729–1738 (2011). Article Google Scholar * Messina, J. L. et al. 12-Chemokine gene signature identifies lymph node-like structures
in melanoma: potential for patient selection for immunotherapy? _Sci. Rep._ 2, 765 (2012). Article Google Scholar * Goc, J. et al. Dendritic cells in tumor-associated tertiary lymphoid
structures signal a TH1 cytotoxic immune contexture and license the positive prognostic value of infiltrating CD8+ T cells. _Cancer Res._ 74, 705–715 (2014). Article CAS Google Scholar *
Behr, D. S. et al. Prognostic value of immune cell infiltration, tertiary lymphoid structures and PD-L1 expression in Merkel cell carcinomas. _Int. J. Clin. Exp. Pathol._ 7, 7610–7621
(2014). PubMed PubMed Central Google Scholar * Caro, G. D. et al. Occurrence of tertiary lymphoid tissue is associated with T-cell infiltration and predicts better prognosis in
early-stage colorectal cancers. _Clin. Cancer Res._ 20, 2147–2158 (2014). Article Google Scholar * Petitprez, F. et al. B cells are associated with survival and immunotherapy response in
sarcoma. _Nature_ 577, 556–560 (2020). Article CAS Google Scholar * Cabrita, R. et al. Tertiary lymphoid structures improve immunotherapy and survival in melanoma. _Nature_ 577, 561–565
(2020). Article CAS Google Scholar * Helmink, B. A. et al. B cells and tertiary lymphoid structures promote immunotherapy response. _Nature_ 577, 549–555 (2020). Article CAS Google
Scholar * Tsou, P., Katayama, H., Ostrin, E. J. & Hanash, S. M. The emerging role of B cells in tumor immunity. _Cancer Res._ 76, 5597–5601 (2016). Article CAS Google Scholar *
Bruno, T. C. New predictors for immunotherapy responses sharpen our view of the tumour microenvironment. _Nature_ 577, 474–476 (2020). Article CAS Google Scholar * Kroeger, D. R., Milne,
K. & Nelson, B. H. Tumor-Infiltrating plasma cells are associated with tertiary lymphoid structures, cytolytic T-cell responses, and superior prognosis in ovarian cancer. _Clin Cancer
Res._ 22, 3005–3015 (2016). Article CAS Google Scholar * Germain, C. et al. Presence of B cells in tertiary lymphoid structures is associated with a protective immunity in patients with
lung cancer. _Am. J. Respir. Crit. Care Med._ 189, 832–844 (2014). Article CAS Google Scholar * Montfort, A. et al. A strong B-cell response is part of the immune landscape in human
high-grade serous ovarian metastases. _Clin. Cancer Res._ 23, 250–262 (2017). Article CAS Google Scholar * Kalergis, A. M. & Ravetch, J. V. Inducing tumor immunity through the
selective engagement of activating Fcγ receptors on dendritic cells. _J. Exp. Med._ 195, 1653–1659 (2002). Article CAS Google Scholar * Fridman, W. H. et al. B cells and cancer: To B or
not to B? _J. Exp. Med._ 218, e20200851 (2020). Article Google Scholar * Lu, Y. et al. Complement signals determine opposite effects of B cells in chemotherapy-induced immunity. _Cell_
180, 1081–1097 (2020). Article CAS Google Scholar * Schwartz, L. H. et al. RECIST 1.1—update and clarification: from the RECIST committee. _Eur. J. Cancer_ 62, 132–137 (2016). Article
Google Scholar * Vigliar, E. et al. PD-L1 expression on routine samples of non-small cell lung cancer: results and critical issues from a 1-year experience of a centralised laboratory. _J.
Clin. Pathol._ 72, 412–417 (2019). Article CAS Google Scholar * Balar, A. V. et al. First-line pembrolizumab in cisplatin-ineligible patients with locally advanced and unresectable or
metastatic urothelial cancer (KEYNOTE-052): a multicentre, single-arm, phase 2 study. _Lancet Oncol._ 18, 1483–1492 (2017). Article CAS Google Scholar * Koeppel, F. et al. Added value of
whole-exome and transcriptome sequencing for clinical molecular screenings of advanced cancer patients with solid tumors. _Cancer J._ 24, 153–162 (2018). Article CAS Google Scholar
Download references ACKNOWLEDGEMENTS This study was funded by AstraZeneca. AUTHOR INFORMATION Author notes * These authors contributed equally: Lucile Vanhersecke, Maxime Brunet, Alban
Bessede, Wolf H. Fridman, François Le Loarer, Antoine Italiano. AUTHORS AND AFFILIATIONS * Department of Pathology, Institut Bergonié, Bordeaux, France Lucile Vanhersecke, Isabelle
Soubeyran, Valérie Velasco & François Le Loarer * Faculty of Medicine, University of Bordeaux, Bordeaux, France Lucile Vanhersecke, Maxime Brunet, Mathieu Larroquette, François Le Loarer
& Antoine Italiano * Department of Medicine, Institut Bergonié, Bordeaux, France Maxime Brunet, Sophie Cousin, Mathieu Larroquette, Maud Toulmonde, Guilhem Roubaud, Simon Pernot,
Mathilde Cabart, François Chomy, Corentin Lefevre, Kevin Bourcier & Antoine Italiano * Explicyte Immuno-Oncology, Bordeaux, France Jean-Philippe Guégan, Christophe Rey, Félicie Courgeon
& Alban Bessede * Centre de Recherche des Cordeliers, INSERM, Sorbonne Université, Equipe Labellisée Ligue Nationale Contre le Cancer, USPC Université de Paris, Paris, France Antoine
Bougouin, Ilenia Giglioli, Catherine Sautès-Fridman & Wolf H. Fridman * Department of Oncology, Clinique Marzet, Pau, France Sylvestre Le Moulec * Department of Medicine, Gustave Roussy,
Villejuif, France Benjamin Besse, Yohann Loriot, Aurélien Marabelle, Jean-Charles Soria & Antoine Italiano * Department of Radiology, Institut Bergonié, Bordeaux, France Michèle Kind *
AstraZeneca, Rahway, NJ, USA Ezoglin Oflazoglu & Ariel Savina * Clinical Research and Clinical Epidemiology Unit (ISO 9001 Certified), Institut Bergonié, Comprehensive Cancer Centre,
Bordeaux, France Carine Bellera & Casimir Sofeu Authors * Lucile Vanhersecke View author publications You can also search for this author inPubMed Google Scholar * Maxime Brunet View
author publications You can also search for this author inPubMed Google Scholar * Jean-Philippe Guégan View author publications You can also search for this author inPubMed Google Scholar *
Christophe Rey View author publications You can also search for this author inPubMed Google Scholar * Antoine Bougouin View author publications You can also search for this author inPubMed
Google Scholar * Sophie Cousin View author publications You can also search for this author inPubMed Google Scholar * Sylvestre Le Moulec View author publications You can also search for
this author inPubMed Google Scholar * Benjamin Besse View author publications You can also search for this author inPubMed Google Scholar * Yohann Loriot View author publications You can
also search for this author inPubMed Google Scholar * Mathieu Larroquette View author publications You can also search for this author inPubMed Google Scholar * Isabelle Soubeyran View
author publications You can also search for this author inPubMed Google Scholar * Maud Toulmonde View author publications You can also search for this author inPubMed Google Scholar *
Guilhem Roubaud View author publications You can also search for this author inPubMed Google Scholar * Simon Pernot View author publications You can also search for this author inPubMed
Google Scholar * Mathilde Cabart View author publications You can also search for this author inPubMed Google Scholar * François Chomy View author publications You can also search for this
author inPubMed Google Scholar * Corentin Lefevre View author publications You can also search for this author inPubMed Google Scholar * Kevin Bourcier View author publications You can also
search for this author inPubMed Google Scholar * Michèle Kind View author publications You can also search for this author inPubMed Google Scholar * Ilenia Giglioli View author publications
You can also search for this author inPubMed Google Scholar * Catherine Sautès-Fridman View author publications You can also search for this author inPubMed Google Scholar * Valérie Velasco
View author publications You can also search for this author inPubMed Google Scholar * Félicie Courgeon View author publications You can also search for this author inPubMed Google Scholar *
Ezoglin Oflazoglu View author publications You can also search for this author inPubMed Google Scholar * Ariel Savina View author publications You can also search for this author inPubMed
Google Scholar * Aurélien Marabelle View author publications You can also search for this author inPubMed Google Scholar * Jean-Charles Soria View author publications You can also search for
this author inPubMed Google Scholar * Carine Bellera View author publications You can also search for this author inPubMed Google Scholar * Casimir Sofeu View author publications You can
also search for this author inPubMed Google Scholar * Alban Bessede View author publications You can also search for this author inPubMed Google Scholar * Wolf H. Fridman View author
publications You can also search for this author inPubMed Google Scholar * François Le Loarer View author publications You can also search for this author inPubMed Google Scholar * Antoine
Italiano View author publications You can also search for this author inPubMed Google Scholar CONTRIBUTIONS A.I., A. Bessede, W.H.F. and F.L.L. conceived of and designed the study. L.V. and
F.L.L. performed the histological analyses. S.C., S.L.M., I.S., M.T., G.R., S.P., M.C., F.C., C.L., K.B., M.K., F.C., B.B., Y.L., A.M., J.-C.S. and A.I. provided study materials or treated
patients. All authors collected and assembled data. A.I., A. Bessede, L.V. and F.L.L. developed the tables and figures. A.I., A. Bessede, M.B., F.L.L., W.H.F. and C.S.-F. conducted the
literature search and wrote the manuscript. All authors were involved in the critical review of the manuscript and approved the final version. CORRESPONDING AUTHOR Correspondence to Antoine
Italiano. ETHICS DECLARATIONS COMPETING INTERESTS A. Bessede, J.-P.G. and C.R. are employees of ImmuSmol/Explicyte. E.O. and A.S. are employees of AstraZeneca. A.I. received research grants
from AstraZeneca, Bayer, Bristol Myers Squibb, Chugai, Merck, MSD, PharmaMar, Novartis and Roche and received personal fees from Epizyme, Bayer, Lilly, Roche and SpringWorks. B.B. received
grants from AstraZeneca, Pfizer, Eli Lilly, Onxeo, Bristol Myers Squibb, Inivata, AbbVie, Amgen, Blueprint Medicines, Celgene, GlaxoSmithKline, Ignyta, Ipsen, Merck KGaA, MSD Oncology,
Nektar, PharmaMar, Sanofi, Spectrum Pharmaceuticals, Takeda, Tiziana Therapeutics, Cristal Therapeutics, Daiichi Sankyo, Janssen Oncology, OSE Immunotherapeutics, BeiGene, Boehringer
Ingelheim, Genentech, Servier and Tolero Pharmaceuticals. Y.L. received grants from Janssen, Sanofi and MSD, personal fees from Janssen, Sanofi, Astellas, Roche, AstraZeneca, Bristol Myers
Squibb, Seattle Genetics, MSD, Clovis, Incyte and Pfizer and non-financial support from Astellas, Roche, AstraZeneca, Bristol Myers Squibb, Seattle Genetics and MSD. A.M. received research
grants from Merus, Bristol Myers Squibb, Boehringer Ingelheim, Transgene and MSD and personal fees from Bristol Myers Squibb, AstraZeneca, MedImmune, Oncovir and Merieux. J.-C.S. received
consultancy fees from AstraZeneca, Astex, Clovis, GlaxoSmithKline, GamaMabs, Lilly, MSD, Mission Therapeutics, Merus, Pfizer, PharmaMar, Pierre Fabre, Roche/Genentech, Sanofi, Servier,
Symphogen and Takeda. L.V., M.B., S.C., S.L.M., M.L., I.S., M.T., G.R., S.P., M.C., F.C., C.L., K.B., M.K., I.G., C.S.-F., V.V., F.C., W.H.F. and F.L.L. declare no competing interests.
ADDITIONAL INFORMATION PEER REVIEW INFORMATION _Nature Cancer_ thanks the anonymous reviewers for their contribution to the peer review of this work. PUBLISHER’S NOTE Springer Nature remains
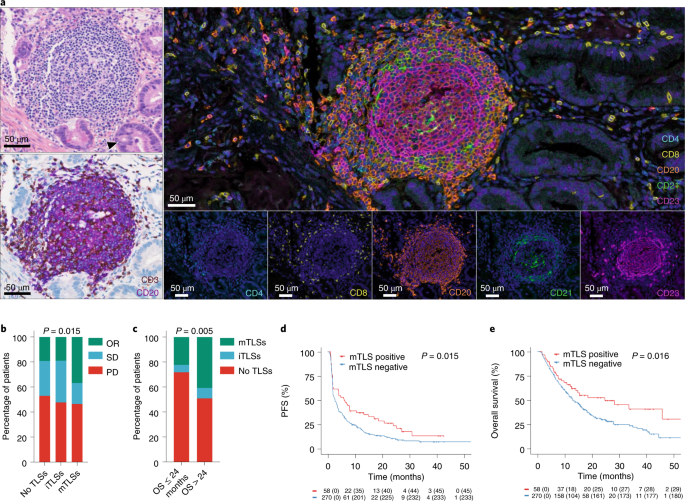
neutral with regard to jurisdictional claims in published maps and institutional affiliations. EXTENDED DATA EXTENDED DATA FIG. 1 ASSESSMENT OF THE PRESENCE OF TLS AND THEIR MATURATION
STAGE IN TUMORS. A: This is a TLS-positive primary pancreatic adenocarcinoma associated with a T CD8 + lymphocyte density of 154/mm² and negative for PD-L1. The TLS are delineated with the
black lines on the HES slide, highlighting their vicinity to tumor cells. Scale bar indicates 300 µm in size. Representative of 540 tumors analyzed (Discovery cohort n = 328, validation
cohort A n = 131, validation cohort B n = 81). B: This panel shows representative examples of immature and mature TLS observed in tumor samples. Upper panel: This mature TLS is detected in a
primary pancreatic adenocarcinoma associated with a T CD8 + lymphocyte density of 154/mm² and negative for PD-L1. Mature TLS are defined by the presence of a network of CD23-positive
dendritic cells on immunofluorescence (note that in this case, the presence of a germinal center visible on the HES was already diagnostic of a mature TLS). Lower panel: The picture shows an
immature TLS detected in a primary adenocarcinoma of the lung concomitantly showing a high T CD8 + lymphocyte infiltrate of 372/mm² and a PD-L1 TPS score of 1%. The tumor was only
associated with immature TLS displaying no germinal center and no network of CD23-positive dendritic cells on immunofluorescence. Representative of 540 tumors analyzed (Discovery cohort n =
328, validation cohort A n = 131, validation cohort B n = 81). The pictures from the left to right column correspond to 1) Hematoxylin Eosin Saffron (HES) staining, 2) Double
immunohistochemistry staining of CD3/CD20 (CD3 in brown, CD20 in purple), 3) Double immunohistochemistry staining of CD8/PD-L1 (CD8 in brown, PD-L1 in purple), 4) Multiplex
immunofluorescence assay of CD4 (blue), CD8 (yellow), CD20 (orange), CD21 (green) and CD23 (pink). The scale bars on the HES images indicate 50 µm and 100 µm for the upper and lower panels,
respectively. Black cropped arrows highlight the tumor cells in the samples. EXTENDED DATA FIG. 2 PREDICTIVE VALUE OF TLS STATUS ACCORDING TO CPS PD-L1 SCORES. Objective response rates (OR:
objective response, SD: stable disease; PD: progressive disease; chi squared (χ²) test) and Kaplan-Meier curves of progression-free survival (log-rank test) and overall survival (log-rank
test) of 328 cancer patients treated with anti-PD1/PD-L1 antagonists according to CPS PD-L1 scores (a: CPS PD-L1 < 1, N = 223 patients; b: CPS PD-L1 ≥ 1, N = 105 patients) and TLS status
(red curve: mature-TLS positive tumors; blue line: mature-TLS negative tumors). Source data EXTENDED DATA FIG. 3 PREDICTIVE VALUE OF TLS STATUS ACCORDING TO T CD8 CELL DENSITY. Objective
Response rates (OR: objective response, SD: stable disease, PD: progressive disease; chi squared (χ²) test) and Kaplan-Meier curves of progression-free (log-rank test) and overall survival
(log-rank test) of 328 cancer patients treated with anti-PD1/PD-L1 antagonists according to T CD8 + cell density (A: low density, N = 165 patients; B: high density, N = 163 patients) and TLS
status (red curve: mature-TLS positive tumors; blue line: mature-TLS negative tumors). Source data EXTENDED DATA FIG. 4 OUTCOME OF CANCER PATIENTS (NON-SMALL CELL LUNG CANCER EXCLUDED)
ACCORDING TO TLS STATUS. A) Objective Response rates (OR: objective response, SD: stable disease, PD: progressive disease; chi squared (χ²) test) and Kaplan-Meier curves (log-rank test) of
progression-free (B) and overall survival (C) of 201 cancer patients (all tumor types except non-small cell lung cancer) treated with anti-PD1/PD-L1 antagonists according to TLS status (red
curve: mature-TLS positive tumors; blue line: mature-TLS negative tumors). Source data EXTENDED DATA FIG. 5 COMPARISON OF THE TLS SCREENING WITH PATHOLOGY AND IMMUNOHISTOCHEMISTRY PATHOLOGY
VERSUS THE SCREENING WITH IMMUNOFLUORESCENCE (METHOD BY PETITPREZ ET AL). First line: conspicuous mature TLS with CD23 + follicular dendritic cells network. Second line: mature TLS defined
by isolated CD23 + cell displaying dendritic morphology. Pictures from the left to right column correspond to 1) Hematoxylin Eosin Saffron (HES) staining, 2) Multiplex immunofluorescence
assay of CD23 (pink) and CD20 (orange), 3) Double immunohistochemistry staining of CD23/CD20 (CD23 in brown, CD20 in red). The scale bars on the HES images indicate 50 µm in size.
Representative of 540 tumors analyzed (Discovery cohort n = 328, validation cohort A n = 131, validation cohort B n = 81). SUPPLEMENTARY INFORMATION SUPPLEMENTARY INFORMATION Supplementary
Tables 1 and 2. REPORTING SUMMARY SUPPLEMENTARY TABLES Supplementary Tables 3–5. SOURCE DATA SOURCE DATA FIG. 1 Statistical source data. SOURCE DATA FIG. 2 Statistical source data. SOURCE
DATA EXTENDED DATA FIG. 2 Statistical source data. SOURCE DATA EXTENDED DATA FIG. 3 Statistical source data. SOURCE DATA EXTENDED DATA FIG. 4 Statistical source data. RIGHTS AND PERMISSIONS
Reprints and permissions ABOUT THIS ARTICLE CITE THIS ARTICLE Vanhersecke, L., Brunet, M., Guégan, JP. _et al._ Mature tertiary lymphoid structures predict immune checkpoint inhibitor
efficacy in solid tumors independently of PD-L1 expression. _Nat Cancer_ 2, 794–802 (2021). https://doi.org/10.1038/s43018-021-00232-6 Download citation * Received: 16 January 2021 *
Accepted: 08 June 2021 * Published: 12 August 2021 * Issue Date: August 2021 * DOI: https://doi.org/10.1038/s43018-021-00232-6 SHARE THIS ARTICLE Anyone you share the following link with
will be able to read this content: Get shareable link Sorry, a shareable link is not currently available for this article. Copy to clipboard Provided by the Springer Nature SharedIt
content-sharing initiative