High dose vitamin d supplementation alters faecal microbiome and predisposes mice to more severe colitis
- Select a language for the TTS:
- UK English Female
- UK English Male
- US English Female
- US English Male
- Australian Female
- Australian Male
- Language selected: (auto detect) - EN
Play all audios:
ABSTRACT Vitamin D has been suggested as a possible adjunctive treatment to ameliorate disease severity in human inflammatory bowel disease. In this study, the effects of diets containing
high (D++, 10,000 IU/kg), moderate (D+, 2,280 IU/kg) or no vitamin D (D−) on the severity of dextran sodium sulphate (DSS) colitis in female C57Bl/6 mice were investigated. The group on high
dose vitamin D (D++) developed the most severe colitis as measured by blinded endoscopic (p < 0.001) and histologic (p < 0.05) assessment, weight loss (p < 0.001), drop in serum
albumin (p = 0.05) and increased expression of colonic TNF-α (p < 0.05). Microbiota analysis of faecal DNA showed that the microbial composition of D++ control mice was more similar to
that of DSS mice. Serum 25(OH)D3 levels reduced by 63% in the D++ group and 23% in the D+ group after 6 days of DSS treatment. Thus, high dose vitamin D supplementation is associated with a
shift to a more inflammatory faecal microbiome and increased susceptibility to colitis, with a fall in circulating vitamin D occurring as a secondary event in response to the inflammatory
process. SIMILAR CONTENT BEING VIEWED BY OTHERS KETOGENIC DIET ALLEVIATES COLITIS BY REDUCTION OF COLONIC GROUP 3 INNATE LYMPHOID CELLS THROUGH ALTERING GUT MICROBIOME Article Open access 23
April 2021 STRONGER GUT MICROBIOME MODULATORY EFFECTS BY POSTBIOTICS THAN PROBIOTICS IN A MOUSE COLITIS MODEL Article Open access 15 November 2022 ORANGE JUICE CONTAINING _PEDIOCOCCUS
ACIDILACTICI_ CE51 MODULATES THE INTESTINAL MICROBIOTA AND REDUCES INDUCED INFLAMMATION IN A MURINE MODEL OF COLITIS Article Open access 28 October 2023 INTRODUCTION Vitamin D is recognized
as a regulator of both innate and adaptive immune responses1, and vitamin D deficiency has been associated with the development of a number of immune mediated disorders including the
inflammatory bowel diseases (IBD), Crohn’s disease (CD) and ulcerative colitis (UC). Several confounding factors, such as reduced sunlight exposure, low dietary intake and reduced intestinal
absorption, limit the ability to draw conclusions about the causality of the observed link between vitamin D deficiency and active IBD. As a result, investigators have turned to mouse
models of IBD such as the dextran sodium sulphate (DSS)-induced model of colitis2. Vitamin D receptor (VDR) knockout (KO), CYP27B1 KO, as well as dietary vitamin D-deficient mouse models are
more susceptible to colitis3,4,5. In an IL-10-KO mouse model, which typically develop spontaneous colitis, administration of 1,25(OH)2D ameliorated the severity of colitis6. Thus, in select
animal models, vitamin D deficiency increases susceptibility to colitis, and restoring vitamin D sufficiency may ameliorate colitis. There are few studies exploring the effect of high
vitamin D levels on immune regulation. Population studies describe a reverse ‘J’ or ‘U’ phenomenon where both vitamin D deficiency and high vitamin D are associated with increased all-cause
and cardiovascular specific mortality7,8,9. Higher vitamin D levels at birth have also been linked to the development of allergy10,11, and genetic analysis within these cohorts identify
epigenetic changes in a number of genes including the thymic stromal lymphopoietin (_TSLP_) gene that may explain the immune mechanism for predisposition to allergy with elevated vitamin D
levels12. The effect of higher vitamin D levels in clinical IBD or animal models of IBD have not been explored. Vitamin D-related changes to gut microbiota are a possible mechanism for
altering susceptibility to colitis. Vitamin D-deficient mice developed elevated bacterial counts in colonic tissue and greater susceptibility to DSS colitis3. Dietary-induced vitamin D
deficiency alters the composition of the faecal microbiome of C57Bl/6 mice, with an increase in the relative quantities of Bacteroidetes, Firmicutes, Actinobacteria, and Gamma-Proteobacteria
in naïve, non-colitic mice13. The effect of vitamin D on microbiota, however, is not limited to the gastrointestinal tract with an inverse correlation between circulating 25(OH)D levels and
_Pseudomonas_ operating taxonomic units (OTU) observed in the lungs of naïve female BALB/c mice14. Thus, vitamin D may regulate the microbiome at different sites and this could be due to
its effect on innate immune responses, in particular the expression of antimicrobial peptides, such as the cathelicidins and β-defensins15. The aim of this study was, therefore, to determine
the effect of diets supplemented with high doses of vitamin D, compared to standard diets sufficient in vitamin D and diets deficient in vitamin D with no supplementation, on the
susceptibility to DSS-induced colitis. Further, we sought to investigate the effect of different doses of vitamin D on the faecal microbiota and how this correlated with susceptibility to
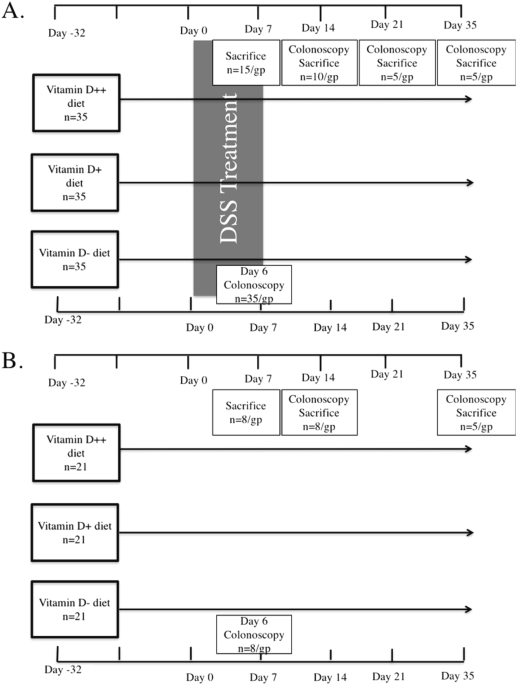
colitis. MATERIALS AND METHODS MICE AND DIET Female 6 week-old C57Bl/6 mice were fed semi-pure diets supplemented with higher than usual doses of vitamin D (SF14-069, Specialty Feeds, Perth,
Western Australia, 10,000 IU/kg vitamin D3, 0.5% calcium), standard doses of vitamin D to achieve vitamin D sufficiency similar to standard chow (SF05-34, Specialty Feeds, 2,280 IU/kg
vitamin D3, 1% calcium) or no added vitamin D to induce vitamin D deficiency (SF05-033, Specialty Feeds, 0 IU/kg vitamin D3, 2% calcium). All experiments were performed according to the
ethical guidelines of the National Health and Medical Research Council of Australia with the approval from the Telethon Kids Institute Animal Ethics Committee (AEC #276). Mice were purchased
from the Animal Resources Centre, Western Australia. Mice were housed under perspex-filtered fluorescent lighting, which emitted no detectable UVB radiation as measured using a UV
radiometer (UVX Digital Radiometer, Ultraviolet Products Inc., Upland, CA, USA). COLITIS MODEL After 4 weeks of being fed the respective diets, colitis was induced by the addition of DSS [3%
(wt/vol)(MP Biomedicals LLC, OH)] to the drinking water for 6 days. Control mice received water without DSS. As the efficacy of DSS varies between batches, the experiments were conducted
using the same batch16. In preliminary experiments, 3% DSS induced adequate colitis with peak weight loss ranging between 0.3% to 10.3% at day 7. Following induction of colitis, mice
recovered over a period of 0–4 weeks without ongoing DSS treatment (Fig. 1). Mouse body weight was assessed daily during DSS treatment and weekly during recovery. The experiment was
repeated, with a total of 35 mice per group. Mice were sacrificed at day 7, 14, 21 and 35. MURINE COLONOSCOPY A high-resolution mouse video endoscopic system was used to assess the level of
colitis. All mice were scoped on day 6 after commencing DSS treatment and then at the time of sacrifice. Mice were anaesthetised using isofluorane unless the colonoscopy was being performed
at the end-point when ketamine 20 mg/ml and xylazine 2 mg/ml by intraperitoneal injection was used. All procedures were digitally recorded then scored in a blinded fashion. The experimental
endoscopy setup consisted of a miniature endoscope (1.9 mm outer diameter), a xenon light source, a triple chip camera, and an air pump (Karl Storz, Germany) to achieve regulated inflation
of the mouse colon. The severity of colitis was determined using the modified ‘Murine Endoscopic Index of Colitis Severity’ (MEICS)16,17. The MEICS system consists of 5 parameters:
thickening of the colon wall, changes of the normal vascular pattern, presence of fibrin, mucosal granularity and stool consistency. Endoscopic grading was performed for each parameter
(scored between 0 and 3) leading to a cumulative score of between 0 (no signs of inflammation) and 15 (endoscopic signs of severe inflammation). Healthy mice had a score of 0–3. UV RADIATION
For UV experiments, a bank of six 40 W lamps (Philips TL UV-B, Eindhoven, The Netherlands) emitting broadband UVR, 250–360 nm, with 65% of the output in the UVB range (280–315 nm), was used
to irradiate mice and to deliver 1 kJ/m2 of UVR onto clean-shaven 8 cm2 dorsal skin. A new sheet of PVC plastic film (0.22 mm) was taped to the top of each Perspex cage immediately before
irradiation to screen wavelengths <290 nm. Sunlamps were held 20 cm above the cages. HISTOLOGICAL ASSESSMENT OF COLITIS Colons were removed with the rectum discarded as this has a
different tissue fibro-structure. The distal 1 cm of colon was dissected, cleaned, formalin-fixed and embedded in paraffin wax. Sections were stained with haemotoxylin and eosin (H&E).
All H&E sections were assessed blindly by a specialist gastroenterological histopathologist (CF) according to the scoring system by Dieleman _et al_.18. In this scoring system, the
severity and depth of inflammation as well as the level of crypt damage and regeneration are scored. MEASUREMENT OF SERUM METABOLITES At the time of sacrifice, blood was drawn by cardiac
puncture. Levels of 25(OH)D3 were measured in the serum by liquid chromatography tandem mass spectroscopy (LC/MS/MS) (Centre for Metabolomics, UWA)19. Levels of 1,25(OH)2D3 were measured
using IDS EIA ELISA kits (Immunodiagnostic Systems Ltd, Fountain Hills, AZ) as described by the manufacturer. The serum vitamin D binding protein (VDBP) concentration was measured in
duplicate using a Quantikine ELISA (R&D Systems, Minneapolis, MN) as per the manufacturer’s instructions. The results were verified by a radial immunodiffusion method as previously
published20. Serum calcium and albumin were measured by standard colorimetric reactions using the Architect c16000 Analyzer (Abbott Diagnositcs, Abbott Park, IL) by PathWest, Royal Perth
Hospital, WA. Serum cytokines were measured using Bio-Plex Pro™ Mouse Cytokine 23-plex panel (Bio-Rad Laboratories, Hercules, CA) as per the manufacturer’s instructions. The cytokines
analysed included interleukin (IL)-1α, IL-1β, IL-2, IL-3, IL-4, IL-5, IL-6, IL-9, IL-10, IL-12 p40, IL-12 p70, IL-13, IL-17, eotaxin (CCL11), G-CSF, GM-CSF, IFN-γ, KC, MCP-1, MIP-1α, MIP-1β,
RANTES and TNF-α. REAL-TIME PCR Messenger RNA was extracted from snap-frozen colon, liver and kidney tissue with cDNA synthesised and real-time assays performed as previously described21.
Real-time PCR primers were _CYP27B1_ cat # 301447280210/0&1, _CYP24A1_ cat # 3014472802-20/0&1, _CYP2R1_ cat # QT0005750 (Sigma-Aldrich co., St. Louis, MO), and for TNFα Qiagen
Quantitect Primer Assay (Qiagen, Hilden, Germany). Housekeeping genes included elongation factor 1α (eEF1α) for kidney and liver tissue, TATA-box-binding protein for colonic tissue
(Sigma-Aldrich co., St. Louis,MO). Quantitect SYBRGreen was used for qPCR on the AB17900HT instrument. Fold-change was determined by using the 2−∆∆Ct method. FAECAL MICROBIOTA ANALYSIS The
faecal microbiome was analysed by sequencing the V3-V4 segment of the 16S ribosomal RNA (rRNA) gene using Illumina MiSeq chemistry. Faecal pellets were collected and stored at −20 °C.
Bacterial DNA was extracted using the PowerSoil® DNA isolation kit according to the manufacturer’s instructions (MO BIO Laboratories, Carlsbad, CA). PCR amplification (341F/806F primer pair)
and sequencing was performed by the Australian Genome Research Facility on the Illumina MiSeq (San Diego, CA) with 2 × 300 bp paired-end chemistry. Paired-ends reads were assembled by
aligning the forward and reverse reads using PEAR (version 0.9.5)22. Primers were trimmed using Seqtk (version 1.0)23. Trimmed sequences were processed using Quantitative Insights into
Microbial Ecology (QIIME 1.8)24 USEARCH (version 7.1.1090)25,26 and UPARSE26 software. Using QIIME, taxonomy was assigned using Greengenes database (Version 13_8, Aug 2013)27. The biom file,
the OTU table, the taxonomic assignments and associated sample data were imported into R to create a phyloseq object. For all beta diversity analyses, OTUs for which the variance across all
samples were very low, were filtered out. For testing a single categorical experimental condition, exact tests for differences in the means between two groups of negative-binomially
distributed counts were computed. Data were normalized using the RLE scaling factor method and dispersions estimated. The counts were extracted and ranked by p-value, applying a false
discovery rate cut-off of less than 0.001. STATISTICAL ANALYSIS Statistical significance was calculated using IBM® SPSS® Statistics Version 22 (IBM Corp. Armonk, NY). All graphs and
comparison of differences between groups were assessed using Student’s unpaired t-test or ANOVA with post hoc LSD analysis for multiple group analysis. Non-parametric data using Mann-Whitney
U and Kruskall-Wallis testing. Microbiome statistical analysis was undertaken using the programming language R, specifically the _phyloseq_ and _edgeR_ packages available through
Bioconductor, a project providing tools for the analysis and comprehension of high-throughput genomic DNA. LEfSe (Linear discriminant analysis effect size) was used to identify
differentially abundant microbial taxa28. RESULTS VITAMIN D3 DIETS Six-week-old C57Bl/6 female mice were fed diets with high (D++), moderate (D+) or no (D−) vitamin D3 for four weeks. At ten
weeks of age, the mice in the three groups were not significantly different in weight (data not shown). D++ mice had significantly higher serum 25(OH)D3 compared to D+ mice which was in
turn higher than D− (Table 1). The serum concentration of activated vitamin D, 1,25(OH)2D3, was not significantly different between the D++ and D+ groups. As previously observed29,
1,25(OH)2D concentrations in the D− mice were lower than in the other groups, though this did not reach statistical significance (p = 0.087). There was no difference in serum calcium levels
between the three groups. VITAMIN D AND DSS-INDUCED COLITIS Body weight loss and liquid stools were observed between 2–7 days after commencing the DSS treatment. All DSS-treated mice lost
weight compared to pre-DSS measures. The peak percentage weight loss occurred at day 7 (Fig. 2A). At day 6 and 7, the D++ mice lost significantly more weight than D+ mice (p < 0.001), and
at day 7 and 8, D− mice lost significantly more weight than D+ mice (p < 0.01) (Fig. 2A,B). At the day 6 colonoscopy, control mice in all groups had solid stool, preserved mucosal
vascularity, normal colonic translucency with a mean MEICS of 0.6 ± 0.17 (n = 23, range 0–3), consistent with no colitis (Fig. 2C). DSS-treated mice demonstrated endoscopic inflammation with
loose stools, loss of intestinal wall translucency and mucosal bleeding with a mean MEICS 4.8 ± 0.2 (n = 102, range 0–10). This was significantly greater than control mice (p < 0.001,
95% CI 3.6–4.7). At day 6, MEICS was significantly greater in D++ (6.3 ± 0.30, n = 33) than in the D+ (4.1 ± 0.30, n = 34, p < 0.001) and D− (3.9 ± 0.33, n = 35, p < 0.001) groups
(Fig. 2C). There was no difference in colitis severity observed in mice from the D+ and D− groups (p = 0.65). At day 14, a higher MEICS was observed in the D++ group compared to the D+ group
(p < 0.01), but this difference resolved by day 21 and 35 as recovery was almost complete (Fig. 2C). The histological grading of colitis at the distal colon on day 14 was greater in mice
from the D++ (8.22 ± 2.53, n = 9) than D+ (1.42 ± 0.57, n = 7, p < 0.05) or D− (1.43 ± 0.57, n = 8, p < 0.05) groups. At Day 7 there was a trend for higher inflammation in D++ and D−
compared to D+ mice though this did not reach statistical significance (p = 0.25) (Supplementary Fig. 1A,B). Among all groups, there was positive correlation between MEICS and day 7 weight
loss (r = 0.60, n = 253, p < 0.001), day 7 histological score (r = 0.51, n = 86, p < 0.001) and day 14 histological score (r = 0.59, n = 56, p < 0.001) (data not shown). At day 7,
the mean serum albumin was less in all groups with colitis (Fig. 2D) than corresponding controls. Among DSS-treated mice, the mean albumin level was significantly lower among the D++,
compared to the D+ (p < 0.05) and D− (p < 0.05) group, consistent with a worse colitis seen in the D++ group. Gene expression of TNF-α in colon tissue by RT-PCR at day 7, was increased
in all DSS-treated groups compared to controls. This was again greatest in the D++ group compared to D− (6.5 ± 3.1 vs 2.2 ± 0.36 fold, p < 0.05) with a trend to be greater than that
measured in the D+ mice (4.1 ± 0.11, p = 0.08) (Fig. 2D). Serum cytokines TNF-α, IFN-γ, IL-10, IL-6, IL-12p40 and IL-1β at day 7 were greater among all DSS mice compared to control mice (p
< 0.05 for all cytokines, data not shown). When comparisons are stratified by vitamin D groups, the DSS group had higher levels than controls though this was not always statistically
significant (Supplementary Fig. 2). IL-12p40 was highest among D++ compared to D− groups (p < 0.01), but similar changes were not seen with other cytokines (Supplementary Fig. 2).
25(OH)D, 1,25(OH)2D AND VDBP CONCENTRATIONS At day 7, serum 25(OH)D reduced by greater than 60% among DSS mice from the D++ group (Fig. 3A). This difference remained at day 35 (p = 0.053, n
= 5). At day 7 a similar but smaller reduction in serum 25(OH)D levels was observed in the D+ group (Fig. 3B). Differences were not seen at the later time points. In mice from the D− group,
given that 25(OH)D levels were already low at baseline, no reduction in 25(OH)D was detectable (Fig. 3C). As the large decrease in 25(OH)D may have been due to an increase in its conversion
to 1,25(OH)2D, changes in the levels of 1,25(OH)2D were investigated. A significant decrease at day 7 in 1,25(OH)2D concentrations (relative to control mice), was detected in both the D++
and D+ groups suggesting that increased conversion was not the cause of 25(OH)D reductions (p < 0.001) (Fig. 3D). As 25(OH)D, and to a lesser extent 1,25(OH)2D, are mostly bound to the
VDBP, we questioned if the large decrease in both these vitamin D metabolites was due to a loss of VDBP. As described previously (Fig. 2D), serum albumin dropped with colitis. Surprisingly,
the VDBP levels, measured by ELISA, increased with colitis in all groups and this was statistically significant in the D++ and D− groups (Fig. 3E). The increase in VDBP with DSS colitis was
also seen when VDBP was measured by radial immunodiffusion, though this was significant among the D+ and D− group and there was a trend to significance among the D++ group (p = 0.076)
(Supplementary Fig. 1C). These data suggest that the induction of colitis increases circulating VDBP levels. KIDNEY _CYP24A1_ GENE EXPRESSION IN DSS COLITIS In an attempt to explain the
reduced 25(OH)D and 1,25(OH)2D at day 7 post-DSS treatment, changes in the level of expression of mRNA of enzymes involved in vitamin D metabolism were explored. Neither liver _CYP2R1_ nor
kidney _CYP27B1_ mRNA levels changed significantly with the induction of colitis (Fig. 4A,B). There was 5.5 ± 1.3 fold more _CYP24A1_ mRNA in the kidneys of DSS-treated mice on D+ diets
compared to their corresponding control group (p < 0.01) (Fig. 4C). Similarly, kidney _CYP24A1_ mRNA was expressed 4.3 ± 0.6 fold more among DSS-treated mice on D− diets compared to
corresponding controls (p < 0.001). Kidney _CYP24A1_ was up-regulated 4.5 ± 0.9 fold among D++ controls compared to D+ controls (p < 0.05) as an appropriate homeostatic mechanism, but
there was no further increase with the induction of colitis. Thus, increased kidney metabolism may help to explain the reduced 25(OH)D and 1,25(OH)2D at day 7 in the D+ group, though a yet
to be identified mechanism must exist to explain the drop in the D++ group. UV RADIATION-INDUCED 25(OH)D AND COLITIS The reduced circulating levels of 25(OH)D and 1,25(OH)2D observed in mice
with DSS-induced colitis could be caused by decreased intestinal absorption of vitamin D. If so, then 25(OH)D derived from skin exposed to UVB radiation should not fall with inflammation.
To test this, mice fed vitamin D-deficient diets for 4 weeks were treated with daily UV radiation (1 kJ/m2) for 4 days followed by biweekly UV (1 kJ/m2) for the remainder of the experiment
(D−UV+). After the 4 days of UV pretreatment, mice were treated with DSS for a further 6 days. After 4 doses of UV irradiation, serum levels of 25(OH)D in mice from the D−UV+ treatment was
58.0 ± 2.49 nmol/L compared to 4.8 ± 0.15 nmol/L among D− mice without UV treatment (D−UV−) (n = 4/group, p < 0.001), Fig. 5A. The 25(OH)D concentrations among D−UV+ mice were similar to
the D+ without UV treatment (D+UV−). After 6 days of DSS treatment, there was no significant difference in endoscopic severity of colitis between vitamin D-deficient mice (D−) exposed (UV+)
or not exposed (UV−) to UV radiation (MEICS 4.3 ± 0.45 vs 3.9 ± 0.33, n = 35/gp, p = 0.42), nor was there a difference compared to D+UV− mice (MEICS 4.3 ± 0.45 vs 4.12 ± 0.45, n = 35/group,
p = 0.64), Supplementary Fig. 1D. By day 7, the 25(OH)D (Fig. 5B) and 1,25(OH)2D concentrations (not shown) were significantly lower among UV-irradiated vitamin D-deficient, DSS-treated mice
as compared to corresponding controls. Thus, these data suggest that the drop in circulating 25(OH)D in mice where vitamin D is acquired only through irradiation of the skin, cannot be due
to malabsorption. EFFECT OF VITAMIN D ON FAECAL MICROBIOTA Microbiota analysis was performed on 42 faecal samples, comprised of 5 samples from each of the vitamin D groups among controls and
DSS mice at day 7 and 4 samples per group among controls at day 35. One control mouse was considered an outlier and excluded from further analyses (Supplementary Fig. 3A). EFFECT OF VITAMIN
D ON FAECAL MICROBIOTA FROM CONTROL, NON-DSS MICE There were no differences seen in α-diversity as measured by species richness, evenness or Shannon’s diversity in day 7 samples collected
from plain water-treated control mice, and the result was reproducible for day 35 samples (Fig. 6A, Supplementary Fig. 3B,C). Similarly, no significant differences in β-diversity were noted
between day 7 and day 35 samples from control mice (data not shown). Further analysis was carried out only on day 7 samples. Comparisons between vitamin D groups found increasing vitamin D
doses did not affect species richness as measured by chao1 among control mice (Fig. 6A), but it did reduce Shannon’s diversity between D++ controls compared to D− controls (Fig. 6B). No
significant difference was noted between D− and D+ control groups; however, PERMANOVA analysis, a measure of global β-diversity, confirmed significant differences between D− and D++ controls
(p = 0.012, t = 1.68, Permutations = 126) and D+ and D++ controls (P = 0.01, t = 1.76, Permutations = 126). To examine the effect of vitamin D grouping on individual taxa, linear
discriminant analysis (LEfSe) was performed. Forty microbial taxa at all taxonomic levels were found to be significantly different between the three vitamin D groups, of which 37 showed
strong associations (linear discriminant analysis score > 3) (Supplementary Table Ia). To determine the effect of measured serum 25(OH)D3 levels on individual taxa, correlation analysis
was measured using distance based linear modelling (DistLM) analysis between serum 25(OH)D3 levels (Euclidean distance resemblance matrix) and relative abundances of microbial taxa. This
identified a significant correlation with four taxa (>0.1% average relative abundance) which included: _Paulidibacter_|OTU46 (Pseudo-F: 4.6, P = 0.04, Df: 26); Bacteroidales|S24-7|OTU58
(Pseudo-F: 6.7, P = 0.02, Df: 26); _Sutterella_|OTU174 (Pseudo-F: 5.1, P = 0.038, Df: 26); and _Coprococcus_|OTU118 (Pseudo-F: 4.8, P = 0.02, Df: 26). To further inform our 25(OH)D3
correlation analyses and establish the response of these four taxa to vitamin D intake, the relative abundance of these four taxa across each vitamin D diet group were plotted (Fig. 6C). The
relative abundance of _Paulidibacter_|OTU46, Bacteroidales|S24-7|OTU58, and _Sutterella_|OTU174 increased with vitamin D intake, while _Coprococcus_|OTU118 decreased. EFFECT OF DSS COLITIS
ON FAECAL MICROBIOTA Treatment with DSS reduced the number of operational taxonomic units (OTUs) within samples analysed at day 7 from the D− (_P_ = 0.09) and D+ (_P_ = 0.04) but not D++
group relative to D− controls (not shown), but there was no significant difference between the DSS groups (Fig. 7A). DSS did not affect other measures of α-diversity, in particular species
evenness and Shannon’s diversity (Fig. 7B, Supplementary Fig. 3D). However, DSS had a significant impact on overall microbial composition (β-diversity) at day 7 (Fig. 7C). Further, 111
microbial taxa at all taxonomic levels were found to be differentially abundant between controls and DSS mice using LEfSe analysis (Supplementary Table Ib). There was enrichment with DSS of
disease-associated Proteobacteria and a reduction in taxa belonging to Firmicutes. Notably, _Sutterella_ OTU174 increased in relative abundance in DSS mice as compared to controls (LDA
score: 4.27, p < 0.0001) (Fig. 7D, Supplementary Table Ia). This is relevant given similar rise of _Sutterella_ in non-DSS treated controls from the D++ group, Fig. 6C, suggesting a shift
in faecal microbiome of D++ controls to that of DSS mice. Further, when examining the overall microbiome composition there is a clear shift for D++ mice towards that of DSS mice (Fig. 7E).
DISCUSSION This is the first study to examine the effect of high dose vitamin D supplementation in an IBD model. High dose vitamin D supplementation led to more severe DSS colitis as
measured by blinded endoscopic and histologic assessment, weight loss and fall in serum albumin. The development of colitis was associated with an acute drop in serum 25(OH)D and 1,25(OH)2D
levels by a mean of 62% among the high vitamin D group and 23% among the vitamin D sufficient group. Other groups have demonstrated a drop in 1,25(OH)2D3 with DSS colitis but not in
25(OH)D35. This is most likely because the change in 25(OH)D3 was most apparent in the high vitamin D group where the baseline 25(OH)D3 level was significantly greater, this diet has not
been examined in the prior studies of DSS colitis. The drop in 25(OH)D3 was associated with a greater than five-fold increase in gene expression of kidney CYP24A1 among D+ mice, an enzyme
responsible for the catabolism of both 25 and 1,25(OH)2D. This suggests that vitamin D metabolites drops in response to inflammation. We did not observe this increase in kidney CYP24A1
expression in the D++ group which had the greatest drop in 25(OH)D, likely due to increased expression at baseline as a counter-regulatory mechanism to the higher 25(OH)D levels. An
alternative mechanism must exist to explain the fall in vitamin D levels in this group. It is well established that 1,25(OH)2D can be produced by colonic epithelial cells30 and monocytes31,
and this is also regulated by local CYP27B1 and CYP24A1. In fact, a study by Liu _et al_., demonstrated increased Cyp24A1 expression from the proximal, but not distal, colon with DSS
colitis15. Thus, it is possible that with the colon inflammation, colonic epithelial cell and monocyte CYP24A1 expression is upregulated leading to the fall in serum 25(OH)D levels. The
recovery of 25(OH)D3 was slow and did not return to baseline by day 35 in the D++ group despite resolution of colitis. This is likely due to this group having the greatest drop in 25(OH)D3,
thus the time taken for 25(OH)D3 to return to baseline would be expected to take the longest even after the colitis has resolved. In critical illness, reduced 25(OH)D and 1,25(OH)2D levels
have been observed, however this has been in association with reduced concentrations of the VDBP32. In our model we observed an increase in VDBP levels. While this could simply be a
discrepancy between mouse and human vitamin D metabolism, it may also be unique to intestinal inflammation. In our previous work examining over 300 patients with CD in remission, high VDBP
concentrations were independently associated with a 20% increased risk of subsequent clinical relapse of disease33. Thus, subclinical inflammation may lead to increased VDBP levels and
subsequent disease flare. VDBP has an important role in actin scavenging such that in the case of acute tissue damage, cellular actin will bind plasma VDBP for subsequent rapid clearance of
the complex34. It is possible that in response to intestinal tissue damage, VDBP was upregulated and thus is a biomarker of subclinical disease activity. Contrary to previous animal studies,
vitamin D deficiency did not predispose mice to worse colitis, though the measurement of colitis severity by endoscopy in this study differed to the clinical measurements reported in other
studies. We did however observe transient weight loss and a trend for increased histological inflammation of the colon in the vitamin D-deficient group consistent with other reports.
Previously reported rodents with increased susceptibility to DSS colitis have included vitamin D receptor- and Cyp27B1- KO mice. However, they had absolute vitamin D deficiency and numerous
complicating skeletal abnormalities such that they may not be a good representation of the human condition4,5. Animal models of dietary-induced vitamin D deficiency better mimic clinical
deficiency observed in humans; however, mixed results have been reported in these vitamin D deficient mice3,35. Serum cytokines, TNF-α, IFN-γ, IL-10, IL-6, IL-12p40 and IL-1β, increased with
the induction of DSS colitis, and is consistent with previous work from our group16. No differences were seen in serum cytokine levels between the different vitamin D groups, with the
exception of IL-12p40 which was higher among colitic mice from the D++ group compared to the vitamin D-deficient group. It is possible that other cytokine levels reached a plateau level in
all DSS groups, making it impossible to detect differences between the different vitamin D dietary groups. We have previously reviewed the effect of vitamin D supplementation _in-vivo_ and
_in-vitro_ on innate and adaptive immune cells responses, particularly with respect to gastrointestinal inflammation36. In summary, vitamin D supplementation can have favourable effects in
preserving intestinal epithelial barrier function, production of anti-microbial peptides, attenuating capacity for antigen presentation, reducing dendritic cell maturation and T-cell
proliferation. The effect of different doses of vitamin D on these responses was not examined in this study, but it would be important in future work to identify those responses that
reversed with high vitamin D supplementation. The mechanism by which high dose vitamin D supplementation increased colitis susceptibility remains unclear, but we did observe that vitamin D
had a potent effect on the microbial composition of faeces from plain-water treated, control mice. Interestingly, the microbial composition of faeces from D++ control mice approached that of
DSS mice, suggesting a shift to a more pro-inflammatory microbiome even before starting DSS treatment. Some of the changes seen across vitamin D categories were confirmed by correlation
with serum 25(OH)D3 levels. Most notably there was a consistent increase in _Sutterella_ spp. This same organism was also enriched in mice with colitis. _Sutterella_ spp. have been reported
to be enriched in human subjects with inflammatory bowel diseases37 and a recent study of faecal microbiota transplantation in ulcerative colitis found recipients with increases in
_Sutterella_ spp. were consistently less likely to respond to the treatment38. Similarly, the group Bacteroidales S24-7 showed a strong positive correlation with 25(OH)D3 levels but was
depleted in the DSS mice. It is possible that the significant bloom in this taxon in D++ mice impacts microbiome stability and, in turn, susceptibility to the effects of DSS, thus
potentially predisposing these mice to more severe colitis. It remains unclear if the observed changes in faecal microbiota are a direct response to changes in vitamin D, or mediated
indirectly through changes in mucosal immune responses. While our findings demonstrate an association between vitamin D dosing, faecal microbiota changes and susceptibility to colitis, we
acknowledge that a specifically designed study would be needed to draw definitive conclusions about cause and effect. In conclusion, high dose vitamin D worsens the severity of murine
colitis induced by DSS, and is associated with distinct changes in microbial composition that may be a direct dietary effect or as a result of dysregulation of the gut mucosal immune
response. Future work needs to further explore the effects of high levels of vitamin D on gut mucosal immunity to better understand if high as well as low vitamin D levels lead to a
dysregulation. REFERENCES * Ghaly, S. & Lawrance, I. The role of vitamin D in gastrointestinal inflammation. Expert review of gastroenterology & hepatology, 1–15,
https://doi.org/10.1586/17474124.2014.925796 (2014). * Okayasu, I. _et al_. A novel method in the induction of reliable experimental acute and chronic ulcerative colitis in mice.
_Gastroenterology_ 98, 694–702 (1990). Article PubMed CAS Google Scholar * Lagishetty, V. _et al_. Vitamin D deficiency in mice impairs colonic antibacterial activity and predisposes to
colitis. _Endocrinology_ 151, 2423–2432, https://doi.org/10.1210/en.2010-0089 (2010). Article PubMed PubMed Central CAS Google Scholar * Ooi, J. H., Li, Y., Rogers, C. J. &
Cantorna, M. T. Vitamin d regulates the gut microbiome and protects mice from dextran sodium sulfate-induced colitis. _J Nutr_ 143, 1679–1686, https://doi.org/10.3945/jn.113.180794 (2013).
Article PubMed PubMed Central CAS Google Scholar * Liu, N. _et al_. Altered endocrine and autocrine metabolism of vitamin D in a mouse model of gastrointestinal inflammation.
_Endocrinology_ 149, 4799–4808, https://doi.org/10.1210/en.2008-0060 (2008). Article PubMed PubMed Central CAS Google Scholar * Cantorna, M. T., Munsick, C., Bemiss, C. & Mahon, B.
D. 1,25-Dihydroxycholecalciferol prevents and ameliorates symptoms of experimental murine inflammatory bowel disease. _J Nutr_ 130, 2648–2652 (2000). Article PubMed CAS Google Scholar *
Melamed, M. L., Michos, E. D., Post, W. & Astor, B. 25-hydroxyvitamin D levels and the risk of mortality in the general population. _Archives of internal medicine_ 168, 1629–1637,
https://doi.org/10.1001/archinte.168.15.1629 (2008). Article PubMed PubMed Central Google Scholar * Jia, X., Aucott, L. S. & McNeill, G. Nutritional status and subsequent all-cause
mortality in men and women aged 75 years or over living in the community. _The British journal of nutrition_ 98, 593–599, https://doi.org/10.1017/S0007114507725163 (2007). Article PubMed
CAS Google Scholar * Visser, M., Deeg, D. J. H., Puts, M. T. E., Seidell, J. C. & Lips, P. Low serum concentrations of 25-hydroxyvitamin D in older persons and the risk of nursing home
admission. _The American journal of clinical nutrition_ 84, 616–622; quiz 671-612 (2006). * Weisse, K. _et al_. Maternal and newborn vitamin D status and its impact on food allergy
development in the German LINA cohort study. _Allergy_ 68, 220–228, https://doi.org/10.1111/all.12081 (2013). Article PubMed CAS Google Scholar * Miyake, Y., Tanaka, K., Okubo, H.,
Sasaki, S. & Arakawa, M. Maternal consumption of dairy products, calcium, and vitamin D during pregnancy and infantile allergic disorders. _Ann Allergy Asthma Immunol_ 113, 82–87,
https://doi.org/10.1016/j.anai.2014.04.023 (2014). Article PubMed CAS Google Scholar * Junge, K. M. _et al_. Increased vitamin D levels at birth and in early infancy increase offspring
allergy risk-evidence for involvement of epigenetic mechanisms. _J Allergy Clin Immunol_ 137, 610–613, https://doi.org/10.1016/j.jaci.2015.06.040 (2016). Article PubMed CAS Google Scholar
* Assa, A. _et al_. Vitamin D deficiency promotes epithelial barrier dysfunction and intestinal inflammation. _J Infect Dis_ 210, 1296–1305, https://doi.org/10.1093/infdis/jiu235 (2014).
Article PubMed CAS Google Scholar * Roggenbuck, M. _et al_. Vitamin D and allergic airway disease shape the murine lung microbiome in a sex-specific manner. _Respir Res_ 17, 116,
https://doi.org/10.1186/s12931-016-0435-3 (2016). Article PubMed PubMed Central CAS Google Scholar * Lagishetty, V. _et al_. 1alpha-hydroxylase and innate immune responses to
25-hydroxyvitamin D in colonic cell lines. _The Journal of steroid biochemistry and molecular biology_ 121, 228–233, https://doi.org/10.1016/j.jsbmb.2010.02.004 (2010). Article PubMed
PubMed Central CAS Google Scholar * Ng, Y.-L. _et al_. Secreted protein acidic and rich in cysteine (SPARC) exacerbates colonic inflammatory symptoms in dextran sodium sulphate-induced
murine colitis. _PLoS One_ 8, e77575, https://doi.org/10.1371/journal.pone.0077575 (2013). Article ADS PubMed PubMed Central CAS Google Scholar * Becker, C. _et al_. _In vivo_ imaging
of colitis and colon cancer development in mice using high resolution chromoendoscopy. _Gut_ 54, 950–954, https://doi.org/10.1136/gut.2004.061283 (2005). Article PubMed PubMed Central
MathSciNet CAS Google Scholar * Dieleman, L. A. _et al_. Chronic experimental colitis induced by dextran sulphate sodium (DSS) is characterized by Th1 and Th2 cytokines. _Clin Exp
Immunol_ 114, 385–391 (1998). Article PubMed PubMed Central CAS Google Scholar * Clarke, M. W., Tuckey, R. C., Gorman, S., Holt, B. & Hart, P. H. Optimized 25-hydroxyvitamin D
analysis using liquid–liquid extraction with 2D separation with LC/MS/MS detection, provides superior precision compared to conventional assays. _Metabolomics_ 9, 1031–1040,
https://doi.org/10.1007/s11306-013-0518-9 (2013). Article CAS Google Scholar * Bouillon, R., van Baelen, H. & de Moor, P. The measurement of the vitamin D-binding protein in human
serum. _The Journal of clinical endocrinology and metabolism_ 45, 225–231, https://doi.org/10.1210/jcem-45-2-225 (1977). Article PubMed CAS Google Scholar * Gorman, S., Judge, M. A.
& Hart, P. H. Topical 1,25-dihydroxyvitamin D3 subverts the priming ability of draining lymph node dendritic cells. _Immunology_ 131, 415–425,
https://doi.org/10.1111/j.1365-2567.2010.03315.x (2010). Article PubMed PubMed Central CAS Google Scholar * Zhang, J., Kobert, K., Flouri, T. & Stamatakis, A. PEAR: a fast and
accurate Illumina Paired-End reAd mergeR. _Bioinformatics_ 30, 614–620, https://doi.org/10.1093/bioinformatics/btt593 (2014). Article PubMed CAS Google Scholar * Li, H. _GIT Hub_,
https://github.com/lh3/seqtk (2016). * Caporaso, J. G. _et al_. QIIME allows analysis of high-throughput community sequencing data. _Nature methods_ 7, 335–336,
https://doi.org/10.1038/nmeth.f.303 (2010). Article PubMed PubMed Central CAS Google Scholar * Edgar, R. C. Search and clustering orders of magnitude faster than BLAST. _Bioinformatics_
26, 2460–2461, https://doi.org/10.1093/bioinformatics/btq461 (2010). Article PubMed CAS Google Scholar * Edgar, R. C., Haas, B. J., Clemente, J. C., Quince, C. & Knight, R. UCHIME
improves sensitivity and speed of chimera detection. _Bioinformatics_ 27, 2194–2200, https://doi.org/10.1093/bioinformatics/btr381 (2011). Article PubMed PubMed Central CAS Google
Scholar * DeSantis, T. Z. _et al_. Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. _Appl Environ Microbiol_ 72, 5069–5072,
https://doi.org/10.1128/AEM.03006-05 (2006). Article PubMed PubMed Central CAS Google Scholar * Segata, N. _et al_. Metagenomic biomarker discovery and explanation. _Genome Biol_ 12,
R60, https://doi.org/10.1186/gb-2011-12-6-r60 (2011). Article PubMed PubMed Central Google Scholar * Gorman, S. & Hart, P. H. The current state of play of rodent models to study the
role of vitamin D in UV-induced immunomodulation. _Photochem Photobiol Sci_ 11, 1788–1796, https://doi.org/10.1039/c2pp25108f (2012). Article PubMed CAS Google Scholar * Lechner, D.,
Kallay, E. & Cross, H. S. 1alpha,25-dihydroxyvitamin D3 downregulates CYP27B1 and induces CYP24A1 in colon cells. _Mol Cell Endocrinol_ 263, 55–64,
https://doi.org/10.1016/j.mce.2006.08.009 (2007). Article PubMed CAS Google Scholar * Rook, G. A. _et al_. Vitamin D3, gamma interferon, and control of proliferation of Mycobacterium
tuberculosis by human monocytes. _Immunology_ 57, 159–163 (1986). PubMed PubMed Central CAS Google Scholar * Van den Berghe, G. _et al_. Bone turnover in prolonged critical illness:
effect of vitamin D. _The Journal of clinical endocrinology and metabolism_ 88, 4623–4632, https://doi.org/10.1210/jc.2003-030358 (2003). Article PubMed CAS Google Scholar * Ghaly, S.
_et al_. High Vitamin D-Binding Protein Concentration, Low Albumin, and Mode of Remission Predict Relapse in Crohn’s Disease. _Inflammatory bowel diseases_ 22, 2456–2464,
https://doi.org/10.1097/MIB.0000000000000894 (2016). Article PubMed Google Scholar * Speeckaert, M. M., Speeckaert, R., van Geel, N. & Delanghe, J. R. Vitamin D binding protein: a
multifunctional protein of clinical importance. _Adv Clin Chem_ 63, 1–57 (2014). Article PubMed CAS Google Scholar * Liu, T. _et al_. Vitamin D treatment attenuates 2,4,6-trinitrobenzene
sulphonic acid (TNBS)-induced colitis but not oxazolone-induced colitis. _Scientific reports_ 6, 32889, https://doi.org/10.1038/srep32889 (2016). Article ADS PubMed PubMed Central CAS
Google Scholar * Ghaly, S. & Lawrance, I. The role of vitamin D in gastrointestinal inflammation. _Expert review of gastroenterology & hepatology_ 8, 909–923,
https://doi.org/10.1586/17474124.2014.925796 (2014). Article CAS Google Scholar * Santoru, M. L. _et al_. Cross sectional evaluation of the gut-microbiome metabolome axis in an Italian
cohort of IBD patients. _Scientific reports_ 7, 9523, https://doi.org/10.1038/s41598-017-10034-5 (2017). Article ADS PubMed PubMed Central CAS Google Scholar * Paramsothy, S. _et al_.
Multidonor intensive faecal microbiota transplantation for active ulcerative colitis: a randomised placebo-controlled trial. _Lancet_ 389, 1218–1228,
https://doi.org/10.1016/S0140-6736(17)30182-4 (2017). Article PubMed Google Scholar Download references ACKNOWLEDGEMENTS The authors would like to thank Dr. Michael Clarke, Centre of
Metabolomics, University of Western Australia for performing the vitamin D analyses, Dr. Mario Taranto, Pathwest, Royal Perth Hospital, WA, Australia for performing calcium and albumin
analysis. NOK acknowledges support from the Cancer Institute NSW. SG acknowledges support from GESA-Ferring IBD Clinician Establishment Award 2015. AUTHOR INFORMATION AUTHORS AND
AFFILIATIONS * Telethon Kids Institute, The University of Western Australia, Perth, WA, Australia Simon Ghaly, Terence McGonigle, Danny Mok, Angela Baird, Borut Klopcic, Shelley Gorman &
Prue H. Hart * School of Medicine and Pharmacology, The University of Western Australia, Perth, WA, Australia Simon Ghaly, Frances Lloyd & Ian C. Lawrance * Department of
Gastroenterology and Hepatology, St. Vincent’s Hospital, Sydney, NSW, Australia Simon Ghaly * School of Medical Sciences, UNSW Sydney, Kensington, NSW, Australia Nadeem O. Kaakoush *
Australian Genome Research Facility, The Walter and Eliza Hall Institute, Parkville, Victoria, Australia Lavinia Gordon * Department of Anatomical Pathology, PathWest, Fiona Stanley
Hospital, Murdoch, WA, Australia Cynthia Forest * Clinical and Experimental Endocrinology, Katholieke Universiteit Leuven, Leuven, Belgium Roger Bouillon * Centre for Inflammatory Bowel
Disease, St. John of God Hospital, Subiaco, WA, Australia Ian C. Lawrance Authors * Simon Ghaly View author publications You can also search for this author inPubMed Google Scholar * Nadeem
O. Kaakoush View author publications You can also search for this author inPubMed Google Scholar * Frances Lloyd View author publications You can also search for this author inPubMed Google
Scholar * Terence McGonigle View author publications You can also search for this author inPubMed Google Scholar * Danny Mok View author publications You can also search for this author
inPubMed Google Scholar * Angela Baird View author publications You can also search for this author inPubMed Google Scholar * Borut Klopcic View author publications You can also search for
this author inPubMed Google Scholar * Lavinia Gordon View author publications You can also search for this author inPubMed Google Scholar * Shelley Gorman View author publications You can
also search for this author inPubMed Google Scholar * Cynthia Forest View author publications You can also search for this author inPubMed Google Scholar * Roger Bouillon View author
publications You can also search for this author inPubMed Google Scholar * Ian C. Lawrance View author publications You can also search for this author inPubMed Google Scholar * Prue H. Hart
View author publications You can also search for this author inPubMed Google Scholar CONTRIBUTIONS P.H.H. and S.G. conceived and conducted the experiments, data analysis and drafting of
manuscript. I.C.L. conceived experiment. R.B., F.L., T.M., D.M., A.B., B.K., S.Go. and C.F. conducted experimental work. N.O.K. and L.G. performed data analysis. All authors reviewed the
manuscript. CORRESPONDING AUTHOR Correspondence to Simon Ghaly. ETHICS DECLARATIONS COMPETING INTERESTS The authors declare no competing interests. ADDITIONAL INFORMATION PUBLISHER'S
NOTE: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations. ELECTRONIC SUPPLEMENTARY MATERIAL SUPPLEMENTARY FIGURES RIGHTS
AND PERMISSIONS OPEN ACCESS This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in
any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The
images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not
included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly
from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/. Reprints and permissions ABOUT THIS ARTICLE CITE THIS ARTICLE Ghaly, S.,
Kaakoush, N.O., Lloyd, F. _et al._ High Dose Vitamin D supplementation alters faecal microbiome and predisposes mice to more severe colitis. _Sci Rep_ 8, 11511 (2018).
https://doi.org/10.1038/s41598-018-29759-y Download citation * Received: 15 December 2017 * Accepted: 18 July 2018 * Published: 31 July 2018 * DOI: https://doi.org/10.1038/s41598-018-29759-y
SHARE THIS ARTICLE Anyone you share the following link with will be able to read this content: Get shareable link Sorry, a shareable link is not currently available for this article. Copy
to clipboard Provided by the Springer Nature SharedIt content-sharing initiative